Difference between revisions of "Cefaly"
(update) |
(update) |
||
| Line 33: | Line 33: | ||
<tr> | <tr> | ||
<th scope="row">Price</th> | <th scope="row">Price</th> | ||
| − | <td>[[Costs:: | + | <td>[[Costs::295]] USD <ref>Cefaly Set on the Cefaly official online-shop: http://www.cefalymedical.com/shop/traitement-migraine/english_a/set-cefaly.html (retrieved Jan 28, 2016)</ref></td> |
</tr> | </tr> | ||
| Line 65: | Line 65: | ||
</table> | </table> | ||
| − | Cefaly is head-mounted wearable developed in purpose to treat migraine. | + | Cefaly is head-mounted wearable developed in purpose to treat and prevent migraine. |
<!-- Very brief description of the technology. This section should contain the main information about the subject. It's good to leave this bit as the last one and use the information provided in greater detail in later sections.--> | <!-- Very brief description of the technology. This section should contain the main information about the subject. It's good to leave this bit as the last one and use the information provided in greater detail in later sections.--> | ||
| Line 71: | Line 71: | ||
== Main characteristics == | == Main characteristics == | ||
<!-- This section should describe the technology in more detail. Here should be information about the used hardware and software, available features, chemical composition and so on, provided that they are available. Second half of this section should offer information on history of the technology. When it was created, unveiled, developed, announced to the public or when it was available to purchase. Anything related to the technology that can be pinpointed to a certain date should be in this section together with relevant commentary.--> | <!-- This section should describe the technology in more detail. Here should be information about the used hardware and software, available features, chemical composition and so on, provided that they are available. Second half of this section should offer information on history of the technology. When it was created, unveiled, developed, announced to the public or when it was available to purchase. Anything related to the technology that can be pinpointed to a certain date should be in this section together with relevant commentary.--> | ||
| + | The Cefaly device consists of the headband with the electrode built in. | ||
| + | |||
| + | The company also offers Cefaly to rent for 49 € per 2-month trial period. This is possible only on condition of paying the 295 €. If customer is dissatisfied, the company will pay him/her back 246 €.<ref>TRYING CEFALY on Cefaly official online-shop: http://www.cefaly.com/en/cefaly-shop (retrieved Jan 28, 2016)</ref> | ||
| + | |||
=== Purpose === | === Purpose === | ||
Revision as of 01:13, 28 January 2016
| CEFALY | |
|---|---|

|
|
| Category | Head-mounted devices |
| Developer | STX-Med |
| Announced | 2014[1] |
| Released | Consumers: 2015[2] |
| Price | 295 USD [3] |
| Weight | 45.4 g [4] |
| Dimensions | mm |
| Controls | smartphone |
| Standalone[5] | |
| http://www.cefalytechnology.com/en/company | |
| http://www.cefalytechnology.com/en/products | |
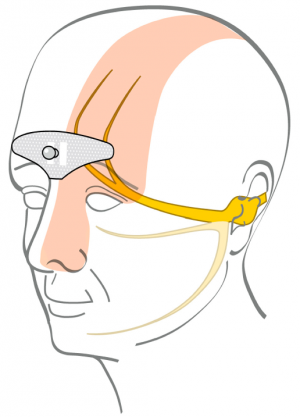
Cefaly is head-mounted wearable developed in purpose to treat and prevent migraine.
Contents
Main characteristics
The Cefaly device consists of the headband with the electrode built in.
The company also offers Cefaly to rent for 49 € per 2-month trial period. This is possible only on condition of paying the 295 €. If customer is dissatisfied, the company will pay him/her back 246 €.[6]
Purpose
Company & People
STX-Med company
Founders:
Pierre Rigaux
Pierre-Yves Muller
Important Dates
2004 - Foundation of STX-Med, which was later named Cefaly.[1]
Ethical Issues
Health Risks
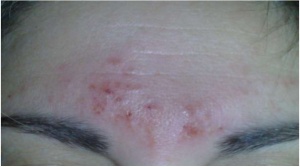
Right on the official page of Cefaly side effects are stated.[2] Company claims, that side effects appear in 4,3% of patients, mentioning most common as intolerance to the feeling of Cefaly on the forehead (1.25%), sensation of fatigue during and after the session (0.65%), headache after one session (0.52%), or irritation of the skin on the forehead (0.22%).
Enhancement/Therapy/Treatment
Public & Media Impact and Presentation
Cefaly has its own official facebook page since 2009, and after this, several pages about Cefaly for specific countries were created.
Public Policy
Announcement about approving migraine treat
Related Technologies, Project or Scientific Research
References
- ↑ FDA approves device to treat migraine headaches: http://articles.latimes.com/2014/mar/11/science/la-sci-sn-fda-approves-device-to-prevent-migraine-headaches-20140311
- ↑ Cefaly Technology release: http://www.biospace.com/News/cefaly-technology-release-10000-u-s-migraine/361631 (retrieved Jan 28, 2016)
- ↑ Cefaly Set on the Cefaly official online-shop: http://www.cefalymedical.com/shop/traitement-migraine/english_a/set-cefaly.html (retrieved Jan 28, 2016)
- ↑ Cefaly® Anti-migrain Device: http://www.amazon.co.uk/Cefaly-Cefaly%C2%AE-Anti-migraine-Device/dp/B009VPAS0M (retrieved Jan 28, 2016)
- ↑ Shows if the device is a standalone wearable computer or if it needs to be connected to a processing unit to function.
- ↑ TRYING CEFALY on Cefaly official online-shop: http://www.cefaly.com/en/cefaly-shop (retrieved Jan 28, 2016)
- ↑ 7.0 7.1 Magis D., Sava S., d’Elia T. S., Baschi R., Schoenen J., Safety and patients’ satisfaction of transcutaneous Supraorbital NeuroStimulation (tSNS) with the Cefaly® device in headache treatment: a survey of 2,313 headache sufferers in the general population http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4177534/